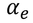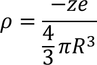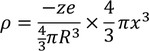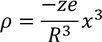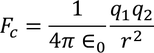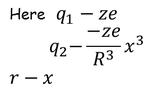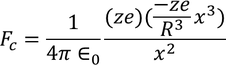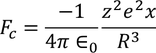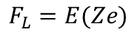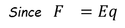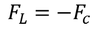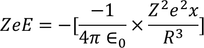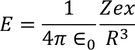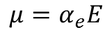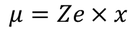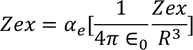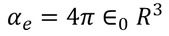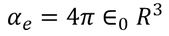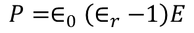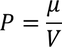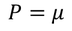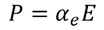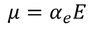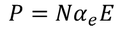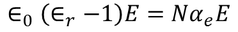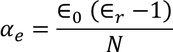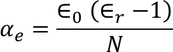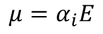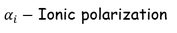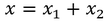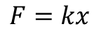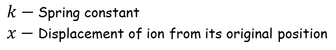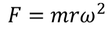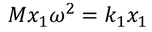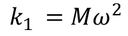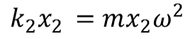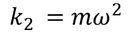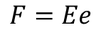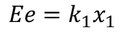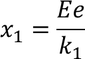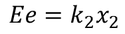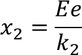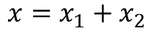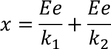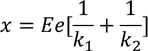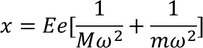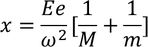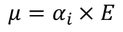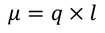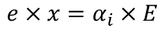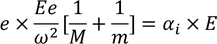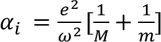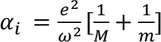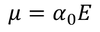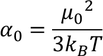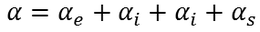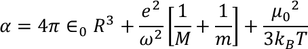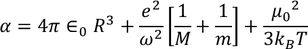Types of polarization:
1. Electronic polarization:
Polarization occurs due to several microscopic mechanisms. When the specimen is placed in DC electric field, it is polarized.
Therefore, The dipole is developed in each atom and the dipole moment is proportional to electric field strength
Therefore, The dipole is developed in each atom and the dipole moment is proportional to electric field strength
- Let us consider an atom of di-electric material is placed in electric field strenght E.
- Assume that charge of nucleus "ze" is surrounding by electron cloud of charge "-ze" is uniformly distributed in atom of radius "R"
- when the field is applied due to large forced nucleus & electron cloud are shifted from their mean positions then coulomb attractive force are created & new equilibrium position will be obtained.
the charge density in sphere of radius R is :
The total -ve charge density. In the sphere of radius x is given by :
The coulomb force of attraction between the nucleus with charge ‘ze’ and the negative charge density of electron cloud
in the sphere of radius x is given by
in the sphere of radius x is given by
Lorentz force of attraction between the nucleus and the electron cloud is
As the system is in equilibrium both the forces are equal and opposite and hence
we know the electric dipole moment
equating both the above equations we get
by solving this equation, we get
Hence the expression for electronic polarizability is
Additional information:
There exist a relation between electronic polarizability and dielectric constant. We know that the polarization P is
There exist a relation between electronic polarizability and dielectric constant. We know that the polarization P is
By the definition polarization P can be written as
consider the volume V=1 then
for N number of electrons the equation changes to
by equating both the polarization values we get
so the relation between polarization and dielectric constant is given by
2. Ionic polarization:
The ionic polarization is due to the displacement of cation and anions when an electric field is applied to a dielectric material. The ionic polarization occurs in ionic solids.
Expression for ionic polarizability:
The electric dipole moment is given by
The electric dipole moment is given by
when an electric field is applied to the ionic crystal positive ions are displaced through distance x1 and negative ions are displaced by the distance x2.
Therefore the total displacement between two ions x is
Therefore the total displacement between two ions x is
Restoring force F is given by
equating both the above equations for positive ion we get,
similarly equating those two forces for negative ion we get,
At equilibrium the Lorentz force is balanced by restoring force in both the ions.
The total distance 'x' is given by
substituting the displacements of the ions in the above equation we get,
Substituting the spring constant values of the ions in the above equation, we get,
We know the electric dipole moment induced is
By the definition of electric dipole moment we have
Equating both the equations we get
So finally ionic polarizability is given by
3. Orientational polarization:
- Orientational polarization is the charecterstic feature of polar dielectric materials.
- In polar dielectric materials, positive and negative charges do not coincide with in the molecule. Hence they possess permanent dipole moment.
- When polar dielectrics are subjected to electric field, dipoles tend to get oriented in the direction of applied field. This phenomenon is called Orientational polarization. The electric dipole moment due to orientation is
Orientational polarizability is given by
This polarization is dependent of temperature. In the case of electronic and ionic polarization, Lorentz force is balanced by restoring force and coulomb attractive force, but for orientational polarization dipole moment is due to thermal agitation of the molecules.
4. space charge polarization:
This polarization occurs with the accumulation of charges with opposite polarity near the electrons.
Total polarization:
Total polarization of a dielectric material = sum of ( electronic polarization, ionic polarization, orientational polarization, space charge polarization)
since the space charge polarization is negligible it is almost taken as zero and
hence the total polarizability is
hence the total polarizability is
Frequency dependence
The effect of polarization depends on time and is given by
- Electronic polarization is very rapid action. This polarization exist when the frequency of the applied voltage is 10^15 Hz
- Ionic polarization is slower than electronic polarization. The frequency range with which ions can be displaced in the infrared region is equal to 10^13 Hz. If the frequency of the applied field is 10^15 Hz the ions do not respond.
- Orientation of dipoles takes comparatively much longer time which is in the range of micro (10^-6) to nano (10^-9) seconds. As a result, the orientational polarization occurs below the micro wave region.