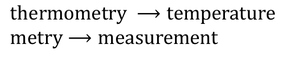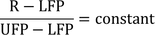Heat :
Heat is a form of energy, transferred between system and its surrounding by virtue of temperature difference.
Thermal equilibrium :
Two bodies are said to be in thermal equilibrium, if no net transfer of heat takes place when they are in contact.
Zeroth law of thermodynamics :
Two bodies A and B are in thermal equilibrium and A and C are in thermal equilibrium then B and C are also in thermal equilibrium.
Heat is a form of energy, transferred between system and its surrounding by virtue of temperature difference.
Thermal equilibrium :
Two bodies are said to be in thermal equilibrium, if no net transfer of heat takes place when they are in contact.
Zeroth law of thermodynamics :
Two bodies A and B are in thermal equilibrium and A and C are in thermal equilibrium then B and C are also in thermal equilibrium.
To understand the above law, there exist a physical quantity, which explains whether two bodies are in thermal equilibrium or not, this physical property is defined as 'Temperature'.
Temperature : Temperature is a physical property of all thermodynamic systems in equilibrium. Two systems are in thermal equilibrium if and only if temperatures are equal.
Its SI unit is kelvin or K.
Its dimensional formula is [θ] or [K]
Temperature : Temperature is a physical property of all thermodynamic systems in equilibrium. Two systems are in thermal equilibrium if and only if temperatures are equal.
Its SI unit is kelvin or K.
Its dimensional formula is [θ] or [K]
Think : Is temperature a scalar?
Thermometry :
Thermometry is the study of measurement of temperature.
A device which measures the condition of a body is called Thermometer.
Measurement of temperature requires
Types of thermometers :
Depending upon thermometric property of a substance, thermometers are classified as below,
To measure temperature, different scales are used. For the definition of any temperature scale, two fixed points are needed. They are
Depending on the numerical values assigned for LFP and UFP, we have different temperature scales. They are
Measurement of temperature requires
- The construction of an instrument called thermometer and
- The calibration of thermometer.
Types of thermometers :
Depending upon thermometric property of a substance, thermometers are classified as below,
- Liquid thermometers are based on the thermal expansion of liquids.
- Gas thermometers are based on the thermal expansion of gas.
- Resistance thermometers are based on the variation of electrical resistance of metals with temperature.
- Thermoelectric thermometers are based on the variation of thermo emf of thermocouple with the temperature difference between the hot and cold junctions of the thermocouple.
- Radiation thermometers are based on the measurement of thermal radiation emitted by a body.
To measure temperature, different scales are used. For the definition of any temperature scale, two fixed points are needed. They are
- Lower fixed point (LFP)
- Upper fixed point (UFP)
Depending on the numerical values assigned for LFP and UFP, we have different temperature scales. They are
- Celsius or Centigrade scale
- Fahrenheit scale
- Kelvin scale and
- Rankine scale
Conversion of temperature from one scale to another :
Since the temperature on different thermometric scale are defined on the basis of the assumption that the liquid expands linearly with temperature, so, if R be the reading of a thermometer corresponding to a certain temperature scale then
Since the temperature on different thermometric scale are defined on the basis of the assumption that the liquid expands linearly with temperature, so, if R be the reading of a thermometer corresponding to a certain temperature scale then
- In the modern thermometry the temperature of triple point of water is ideal fixed point and the other fixed point is the absolute temperature.