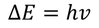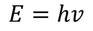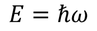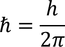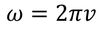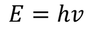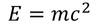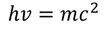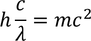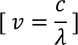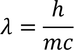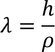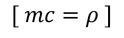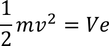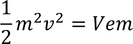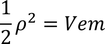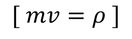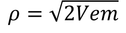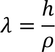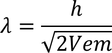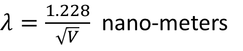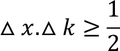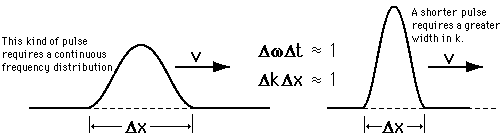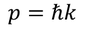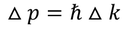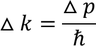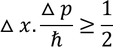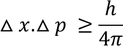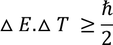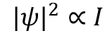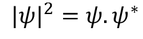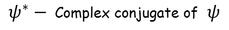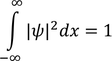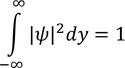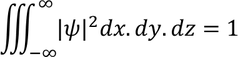Planks quantum theory :
An electron can emit or absorb energy only by making a transition from one energy level to another.
einstein extended Plank's assumptions and stated that electro magnetic radiation is called a photon & each photon of energy
This equation is modified to
Debrolie's hypothesis
When particles are accelerated then they will spread like a wave with certain wave length.
by equating Planks quantum energy and Einstien mass energy, we get,
Heisenberg Uncertainty principleStatement: It is impossible to determine the position and momentum of a moving particle simultaneously with high accuracy in terms of error in position and error in propagation constant
If the error in position is measured accurately then the error in propagation constant becomes less accurate and vice versa.
Let us consider, By substituting this equation in Eq - (1) we get
It can be also written as
In terms of energy and time, the uncertainty principle can be written as
|
Wave FuntionPhysical significance of wave function :Wave function is represented as "ψ"
* All these equations are called Normalization wave equations.
Limitations of wave function:
|