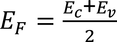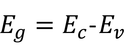SEMI CONDUCTORS
Introduction
- Semiconductors are the materials , whose electronic properties are intermediate between those of good and conductors and insulators.
- Unlike metals a semiconductors has both +ve and -ve carrieres of electricity.
- Germanium and Silicon belongs to IV group are the best examples of semiconductors.
- At 0K all the semiconductors are the insulators.
- Semiconductors have -ve temperature coefficient of resistance.
- The bandgap of semiconductors varies from 0.2 to 3 eV
- Semiconductors are classified into two types.
- Intrinsic semiconductors
- Extrinsic semiconductors
INTRINSIC SEMICONDUCTORS
The pure form of semiconductor is called an Intrinsic semiconductor. Let us consider the case of Germanium , its atomic number is 32 , 2 electrons are in 1st orbit ,8 are in the 2nd orbit ,18 are in the 3rd orbit and remaining 4 are in the outer most orbit.
- Germanium is a trivalent element , each of 4 valency electrons in Germanium atom is shared by valency electrons of 4 adjacent Germanium atoms and makes 4 electron pairs. These types of electron bond are called covalent bonds. A pure Germanium crystal is an insulator at absolute temperature.
- With the increase in temperature a few covalent bonds are broken. Now one electron becomes free by forming a valency known as Hole.
- The hole acts as positively charged particle so it attracts the electrons from the neighbouring atoms to fill the vacancy.
- Thus the hole is shifted to another place of the lattice and it goes on moving from one place to other by attracting electron from neighbouring atoms, through out the crystal.
- On applying electric field, holes move in a direction opposite to that of valency electrons.
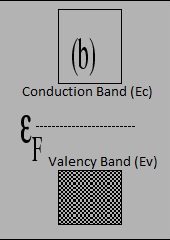
- As the number of charge carriers are equal, fermi energy level lies exactly at the middle of the energy gap.
EXTRINSIC SEMICONDUCTORS
These are impure semiconductors as they are doped with impurities inorder to improve its conductivity. There are two types of extrinsic semiconductors.
- n-type semiconductors
- p-type semiconductors
n-type semiconductor :
- A semiconductor doped with pentavalent impurities is called n-type semiconductor. (examples for pentavalent impurities are Arsenic (Z=33), Antimony (Z=51) etc. )
- When these impurities are added to pure Germanium crystal, four valency electrons of doping material form covalent bonds with four valency of electron of Germanium atom.
- The 5th electron of doping material has no electron to form covalent bond so it remains free and moves randomly in the crystal.
- Each impurity atom donates a free electron to the semiconductor. Hence it as called as " Donor impurity ".
- The energy required to move an electron from donor impurity band to conduction band is 0.127 eV.
- In the case of n-type semiconductors, electrons are the majority charge carriers and holes are minority charge carriers.
p-type semiconductors :
- A semiconductor doped with trivalent impurity is called p-type semiconductor. ( examples for trivalent impurities are Gallium(Z=31) , Indium(Z=49) etc. )
- When a small amount of Indium is added to Germanium crystal, three valence electrons of doped atom forms covalent bonds with three valence electrons of Germanium. There is a deficiency of one electron to complete the fourth bond. This deficiency is called Hole and it carries positive positive charge.
- Hole has a strong tendency to attract an electron so that the valency is shifted to another place. Thus, a hole moves through out the crystal.
- The trivalent impurity is called "Acceptor impurity".
- As the concentration of holes are more, fermi energy level shifts towards valency bond.
- In p-type semiconductor, holes are majority charge carriers and elect are minority charge carriers
Intrinsic semiconductors1. Intrinsic semiconductor is pure material
2. At normal temperature the conductivity is low.
3. The electrons and holes are in equal number.
4. Fermi energy level lies exactly at the center of energy gap.
5. Hall coefficient is always negative.
|
Extrinsic semiconductors1. It is an impure material obtained by adding third or fifth group elements as dopant into an intrinsic semiconductor.
2. At the normal temperature the conductivity is high.
3. In p-type semiconductors, holes are majority charge carriers in n-type electrons are majority charge carriers.
4. In n-type Fermi energy level is close to conduction band and in p-type Fermi energy level is close to valance band .
5. Hall coefficient is negative for electron and it is positive for holes.
|