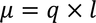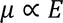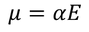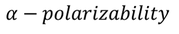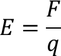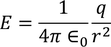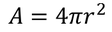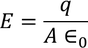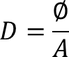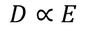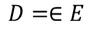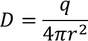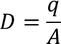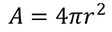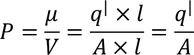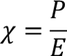Dielectric materials
Introduction:
- All dielectric materials are electrically insulators.
- In dielectrics the electrons are tightly bounded to the nucleus of the atom and there are no free electrons.
- The examples are Mica, Glass, Porcelain, Plastic, Rubber, Wood and Wax.
- All the dielectric materials are insulators but not all insulators are dielectrics.
- Faraday first realized the importance of the dielectrics.
- He introduced the dielectric material between two parallel plate capacitor and found that the charge on the capacitor increases.
Some important definitions:
1. Electric dipole:
The system or the arrangement in which two equal and opposite charges separated through a distance of 'l' is called Electric dipole.
2. Electric dipole moment:
It is the product of magnitude of any one of the charges and the distance between the two charges.
The system or the arrangement in which two equal and opposite charges separated through a distance of 'l' is called Electric dipole.
2. Electric dipole moment:
It is the product of magnitude of any one of the charges and the distance between the two charges.
Note :
The electric dipole moment induced in the atom is proportional to the applied electric field 'E'.
The electric dipole moment induced in the atom is proportional to the applied electric field 'E'.
3. Electric field intensity E:
It is the force experienced by the unit positive charge when it is placed at a point in the field. Units are Newton/Coloumb
It is the force experienced by the unit positive charge when it is placed at a point in the field. Units are Newton/Coloumb
4. Electric flux density:
The Electric flux per unit area is defined as the Electric flux density.
Electric flux density is numerically equals to the surface charge density.
The Electric flux per unit area is defined as the Electric flux density.
Electric flux density is numerically equals to the surface charge density.
We know the value of Electric field intensity E and by substituting we get,
5. Electric Polarization:
The Electric polarization is defined as electric dipole moment per unit volume.
The Electric polarization is defined as electric dipole moment per unit volume.
6. Electric susceptibility:
It is the ratio of electric polarization to the electric field intensity.
It is the ratio of electric polarization to the electric field intensity.
Classification of Dielectrics:
Based on the crystal structure dielectrics are classified into two types.
1. Non-polar Dielectrics:
The dielectric material in which the center of gravity of positive charges and the center of gravity of negative charges coincides with each other are called Non-polar Dielectrics.
The dielectric materials in which the center of gravity of positive charges does not coincide with the center of gravity of negative charges are called Polar dielectric materials.
1. Non-polar Dielectrics:
The dielectric material in which the center of gravity of positive charges and the center of gravity of negative charges coincides with each other are called Non-polar Dielectrics.
- These dielectrics are polarized only when they are placed in electric field and they possess zero dipole moment in the absence of electric field. Examples are Nitrogen, Hydrogen, Benzene.
The dielectric materials in which the center of gravity of positive charges does not coincide with the center of gravity of negative charges are called Polar dielectric materials.
- They have asymmetric structure and possess permanent dipole moment. Examples are Water, Nitrous Oxide.