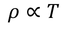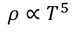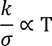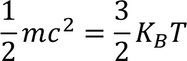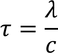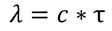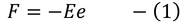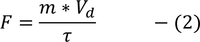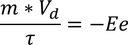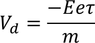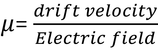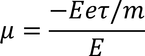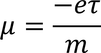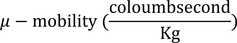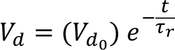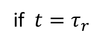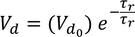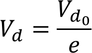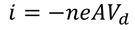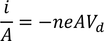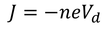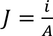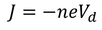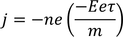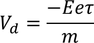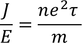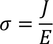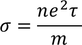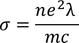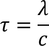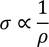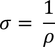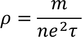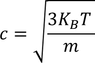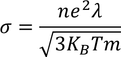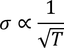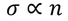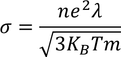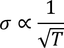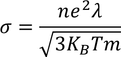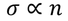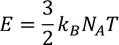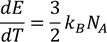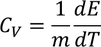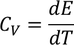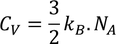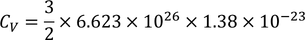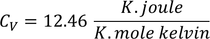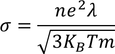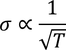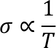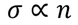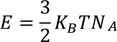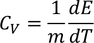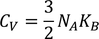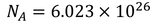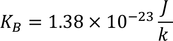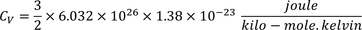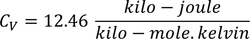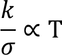Properties of metal :
- All metals obey ohms law
- Metals have high thermal conductivity & electrical conductivity
- Metals obey positive temperature co-efficient of resistance(i.e., as temperature increases resistance increases)
- At room temperature resistivity is proportional to absolute temperature(T)
- At low temperature resistance of metal is directly proportional to 5th power of its absolute temperature(T)
- The ratio of thermal conduction to electrical conduction is directly proportional to absolute temperature
free electron theory of metals by Lorentz- Drude :
- This theory was developed by Lorentz & Drude in 1900
- In metals there are large number of free electrons moving freely in all possible directions
- free electrons in the metals are assumed to behave like gas molecules obeying the kinetic theory of gases
- In metals electrons move randomly and collide with either +ve ions or with free electrons all the collisions are elastic (i.e., no lose of energy)
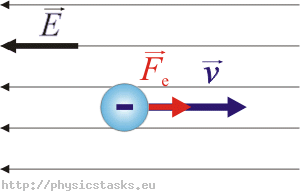
- When electron field is applied to the metal the free electrons accelerated in the opposite direction to the applied field
- The additional velocity acquired by the free electrons in the direction opposite to the applied field is called Drift Velocity
Some important definitionsMean Collision Time :The duration between two successive collisions when electron is in random motion is called Mean collision time
Mean Free Path :The average distance travelled by an electron between two successive collisions during their random motions is called mean free path
Drift Velocity :When an electric field (E) is applied to the metal electrons experience force (F) in opposite direction to the applied field. this force is called Drift Velocity
we know that
(E - Field applied)
(e - electron charge) here -ve sign indicated flow of electrons in opposite direction to the applied field we know also that the force due to its motion
by equating (1) & (2)
Mobility :In steady state the drift velocity per unit electric field is called Mobility.
Relaxation Time :The time taken for the drift velocity of an electron to decay 1/e times of its initial value is called Relaxation Time
|
|
the drift velocity of the electron in the presence of electric field varies as
where electric field is removed, drift velocity decreases gradually
Expression for electric conductionCurrent (i) flowing through a wire having "n" number of electrons and area of cross section "A" is calculated by
Therefore, the relation between current density and drift velocity is given by
Note : Electrical conductance = Current density / Unit electric field
by substituting J/E value in above equation we get
|
Drawbacks of classical free electron theory of metals:
1. Temperature dependence of electrical conductivity:
Hence, Classical free electron fails to explain the temperature dependence of electrical conductivity
2. Electrical conductivity dependence on concentration of electrons:
As per the experimental results there is no direct relation between electrical conductivity and concentration of electrons
3. Specific heat of metal:
If you consider one kilo mole of solid then,
If you consider one kilo mole of solid then,
Equating both Eq - (1) & Eq - (2) we get,
Experimental value is 100 times less than classical value. Hence, Lorentz -Drude theory fails to explain specific heat of a solid.
4. Classical theory fails to explain Wiedemann-franz law that is at lower temperatures, the ratio of thermal conductivity to electrical conductivity is proportional to temperature.
5. Electrical conductivity for semiconductors and insulators are not explained.
5. Electrical conductivity for semiconductors and insulators are not explained.
To be verified (important)According to classical free electron theory
From this equation it is clear that
But according to experimental results
Classical free electron theory fails to explain temperature dependence of electron conductivity
2. electrical condutivity dependence on concentration of electrons :
According to this theory
But as per experimental results, there is no direct relation between electrical conductivity and concentration of electrons
3. Specific heat of metal :
If you considered 1 K.mole of metal
According to the definition of specific heat
by substituting m = 1/K.mole , we get
we know
by substituting these values
Experimental value is 100 times less than classical value. Hence Lawrenge drude fails to explain specific heat of solid
4. It Fails to explain Widemann Frenze law
5. It couldn't explain :
Photo electric effect. Crompton Effect. Black body radiation. Electrical consuctivity for semi-consuctors and insulators |