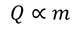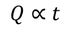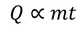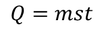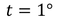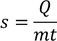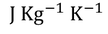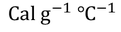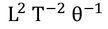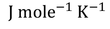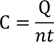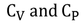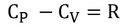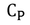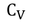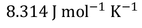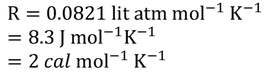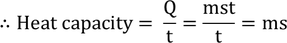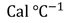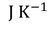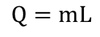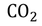The study of measurement of heat is called 'Calorimetry' .
When an amount of heat is supplied to a system, it may
a) Change the temperature of the system (or)
b) Change the state (or)
c) be converted into mechanical work.
Let us consider the case where heat changes the temperature of the body.
When an amount of heat is supplied to a system, it may
a) Change the temperature of the system (or)
b) Change the state (or)
c) be converted into mechanical work.
Let us consider the case where heat changes the temperature of the body.
Heat supplied to a body :
The amount of heat supplied to a body (Q) is directly proportional to its mass (m) to obtain a certain change of temperature.
Similarly Q is directly proportional to change of temperature (t) for certain amount of mass.
then Q=s (numerically)
and here 's' can be called as ' Specific heat capacity '
Specific heat capacity : Specific heat capacity can be defined as " the amount of heat required to raise the temperature of unit mass through one unit "
Specific heat can be expressed as
and here 's' can be called as ' Specific heat capacity '
Specific heat capacity : Specific heat capacity can be defined as " the amount of heat required to raise the temperature of unit mass through one unit "
Specific heat can be expressed as
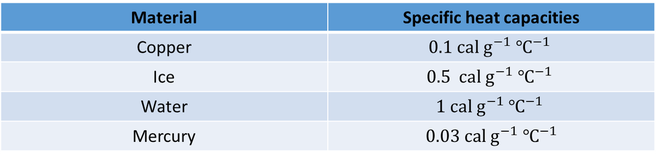
- Specific heat capacities of some most commonly used substances.
- Specific heat capacities can be obtained under different conditions. Most commonly used conditions are constant pressure and constant volume.
- For solids and liquids, Specific heat capacity at constant pressure and specific heat capacity at constant volume have negligible difference.
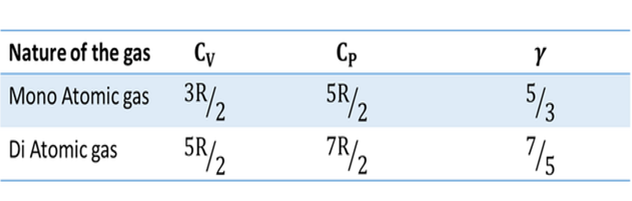
Molar specific heat capacity (C) :
Gases are generally expressed in terms of 'Moles'. Then it is convenient to use molar specific heat capacity.
Molar specific heat capacity can be defined as " The amount of heat required to raise the temperature of one mole of gas through 1 unit ".
Molar specific heat capacity can be defined as " The amount of heat required to raise the temperature of one mole of gas through 1 unit ".
Where 'R' is called Universal gas constant.
The most commonly used Universal gas constant values are
Thermal capacity : The heat required to raise the temperature of a substance through by one degree is known as its Heat or Thermal capacity
Water equivalent : The amount of water whose temperature is raised through one degree by the same quantity of heat which is required to raise the temperature of a body through one degree raise is known as the Water equivalent of the body.
Therefore, Water equivalent of the body = w = ms gram
Therefore, Water equivalent of the body = w = ms gram
Change of state : The transformation of a substance from one state to another is known as change of state.
Melting : When heat is supplied to a solid body at a constant pressure, its temperature gradually rises. After a definite temperature is reached the solid starts to melt. That is at a definite temperature and at a definite pressure, the solid is converted into liquid. The process of attaining the liquid state from solid state is known as Melting. The definite temperature at which melting takes place is known as Melting point
Freezing : If heat is extracted from a liquid at constant pressure, the liquid begins to solidify after attaining the solid state from the liquid state is known as Freezing.
During change of state temperature of the substance remains constant and average distance between the molecules changes.
Latent heat : Heat required to convert unit mass of a substance from solid to liquid state or liquid to gaseous state (at constant pressure) without changing the temperature is called Latent heat of that substance.
Melting : When heat is supplied to a solid body at a constant pressure, its temperature gradually rises. After a definite temperature is reached the solid starts to melt. That is at a definite temperature and at a definite pressure, the solid is converted into liquid. The process of attaining the liquid state from solid state is known as Melting. The definite temperature at which melting takes place is known as Melting point
Freezing : If heat is extracted from a liquid at constant pressure, the liquid begins to solidify after attaining the solid state from the liquid state is known as Freezing.
During change of state temperature of the substance remains constant and average distance between the molecules changes.
Latent heat : Heat required to convert unit mass of a substance from solid to liquid state or liquid to gaseous state (at constant pressure) without changing the temperature is called Latent heat of that substance.
There are two kinds of latent heat
- Latent heat of fusion
- Latent heat of vaporization
Latent heat of fusion : Heat required to convert unit mass of a solid to liquid state without changing its temperature is called the latent heat of fusion of that solid.
The latent heat of fusion of ice is 80 cal/g
Late heat of vaporization : Heat required to convert unit mass of a liquid to vapour without changing its pressure and temperature is called the latent heat of vaporization of that liquid.
Latent heat of vaporization of water is 540 cal/g
Vaporization : The process of conversion of a liquid to the vapour state is known as Vaporization. The state that a liquid attains on vaporization is called the vapour of that particular liquid.
Vaporization of two types
Evaporation occurs from the surface of the liquid. The rate of evaporation increases if the exposed area is large.
Boiling : The process of conversion of a liquid to gaseous state under constant temperature and pressure is known as Boiling. The definite temperature at which boiling takes place is known as Boiling point.
Effects of pressure on melting : The melting point can be raised or lowered by changing pressure.
When pressure is increased, the melting point increases in case of solids such as wax, copper etc. Whose volume increases during the state of conversion from solid to liquid.
Eg : wax, copper etc.
When pressure is increased, the melting point decreases in case of solids whose volume decreases on melting.
Ex : Ice, Bismuth, Antimony etc.
Regelation : The phenomenon that ice melt on application of pressure and again solidifies when pressure is released, is known as Regelation
Change of boiling point with pressure : The boiling point of water is 100℃ under normal atmospheric pressure. If the pressure is lowered below normal atmospheric pressure water will boil at a temperature less than 100℃ . If the pressure is increased above normal atmospheric water will boil at a temperature greater than 100℃
Principle of calorimetry :
Objects at different temperature are made to come in contact with each other on the calorimeter. As a result, heat is exchanged between the objects as well as with the calorimeter. Neglecting any heat exchange with the surroundings, the principle of calorimetry is
" The total heat given by the hot objects equals to total heat received by the cold objects. "
The specific heat capacity of an object can be determined by the following methods.
When a liquid and its vapour remains together in equilibrium the vapour is saturated. The graph plotted between saturation vapour pressure and temperature is known as Phase diagram.
Here point A represents vapour saturation point that means liquid and vapour state co-exist at this case.
The latent heat of fusion of ice is 80 cal/g
Late heat of vaporization : Heat required to convert unit mass of a liquid to vapour without changing its pressure and temperature is called the latent heat of vaporization of that liquid.
Latent heat of vaporization of water is 540 cal/g
Vaporization : The process of conversion of a liquid to the vapour state is known as Vaporization. The state that a liquid attains on vaporization is called the vapour of that particular liquid.
Vaporization of two types
- Evaporation
- Boiling
Evaporation occurs from the surface of the liquid. The rate of evaporation increases if the exposed area is large.
Boiling : The process of conversion of a liquid to gaseous state under constant temperature and pressure is known as Boiling. The definite temperature at which boiling takes place is known as Boiling point.
Effects of pressure on melting : The melting point can be raised or lowered by changing pressure.
When pressure is increased, the melting point increases in case of solids such as wax, copper etc. Whose volume increases during the state of conversion from solid to liquid.
Eg : wax, copper etc.
When pressure is increased, the melting point decreases in case of solids whose volume decreases on melting.
Ex : Ice, Bismuth, Antimony etc.
Regelation : The phenomenon that ice melt on application of pressure and again solidifies when pressure is released, is known as Regelation
Change of boiling point with pressure : The boiling point of water is 100℃ under normal atmospheric pressure. If the pressure is lowered below normal atmospheric pressure water will boil at a temperature less than 100℃ . If the pressure is increased above normal atmospheric water will boil at a temperature greater than 100℃
Principle of calorimetry :
Objects at different temperature are made to come in contact with each other on the calorimeter. As a result, heat is exchanged between the objects as well as with the calorimeter. Neglecting any heat exchange with the surroundings, the principle of calorimetry is
" The total heat given by the hot objects equals to total heat received by the cold objects. "
The specific heat capacity of an object can be determined by the following methods.
- Method of mixtures.
- Newton's law of cooling.
- Electrical method.
When a liquid and its vapour remains together in equilibrium the vapour is saturated. The graph plotted between saturation vapour pressure and temperature is known as Phase diagram.
Here point A represents vapour saturation point that means liquid and vapour state co-exist at this case.
The liquid phase and vapour phase can co-exist along the curve PA. At the points above the curve a pure liquid can exist in equilibrium and at points below this curve a pure vapour exist in equilibrium.
The curve PB represents solid-liquid transition and PC represents solid-vapour transition. These curves also represents, respectively the melting point as a function of pressure, sublimation point as a function of pressure.
These three curves meet at one point labelled P. At the pressure and temperature corresponding to this point, all the three phases may remain together in equilibrium. Then P is known as Triple point.
Triple point of water is at
Pressure = 0.458 cm of Hg,
Temperature = 273.16 K
These three curves meet at one point labelled P. At the pressure and temperature corresponding to this point, all the three phases may remain together in equilibrium. Then P is known as Triple point.
Triple point of water is at
Pressure = 0.458 cm of Hg,
Temperature = 273.16 K
Pressure = 5.11 atm
Temperature = 216.55 K
Temperature = 216.55 K
Eg : In Antarctica one can see the ice bergs are floating on the sea water.